Description
INTRODUCTION: Human respiratory syncytial virus (RSV) is the most common causative agent of serious respiratory tract infections in infants and young children. RSV is an enveloped, non-segmented, negative-sense, single-stranded RNA virus of approximately 15,000 nucleotides that is classified in the genus Orthopneumovirus belonging to the family of Pneumoviridae. RSV infections are often observed during seasonal outbreaks in winter. The virus usually causes a self-limiting upper respiratory tract (URT) infection, however, in a minority of cases, a severe lower respiratory tract (LRT) infection can occur, resulting in pneumonia or bronchiolitis. Current treatment options are limited, and no RSV vaccines are currently licensed. Two major subgroups (A and B) have been identified based on antigenic differences in the attachment G protein. The two subgroups cocirculate during, or alternate between, yearly epidemics and cause indistinguishable disease. Antigenic variation between and within the subgroups may contribute to reinfections with these viruses by evading the host immune responses.
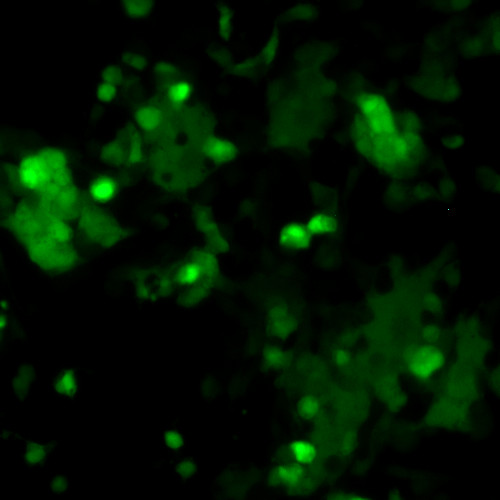
DESCRIPTION: Most of the available information and reagents for RSV are for subgroup A. Here a replication-competent recombinant RSV subgroup B strain expressing green fluorescence protein (GFP) was created using a reverse genetics system. The virus backbone was derived from wildtype RSV strain B1 (GenBank: AF013254.1) of antigenic subgroup B. A GFP gene encoding the enhanced green fluorescence protein was inserted as an additional gene between NS2 and N genes (at the 3rd gene position). Since the sequence of the wild type G gene was refractory to cloning into full-length antigenomic cDNA in E. coli, a codon optimized version of the G gene was incorporated. The resulting virus replicates to wildtype titers in cell culture and efficiently expresses GFP in infected cells.
| Parental Strain: | Subgroup B strain B1 |
| Construction: | GFP gene was inserted between NS2 and N genes as the 3rd gene. |
| Passage History: | The virus was propagated in HEp-2 cells (ATCC CCL-23). |
| Infectivity: | Titer >or= 5.7 log 10 TCID50 per mL. |
| Volume/Storage: | 2 x 1.2 mL per cryovial. Store at –80ºC. |
| Quality Testing: | No bacteria, fungus, or mycoplasma detected. Endotoxin <10 EU/mL. |
| Availability: | Bulk quantity and custom orders are available. Contact info@viratree.com. |
Product Sheet: Product_R423
Certificate of Analysis: CoA_Lot-2409
Material Safety Data Sheet (MSDS): MSDS_RSV